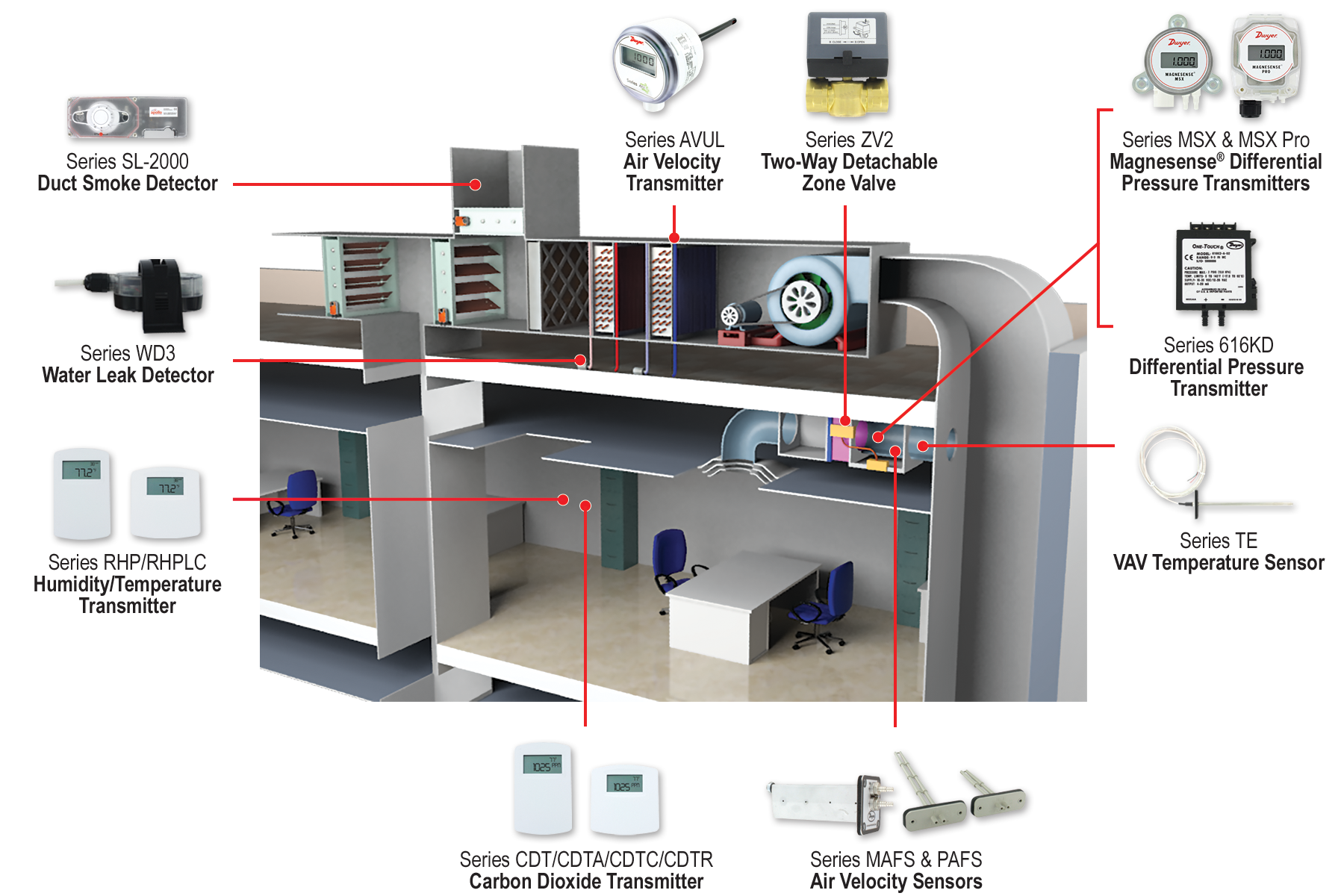
Picture this: you walk into an office building on a hot summer day. As the door closes behind you, you immediately feel the temperature change. It’s cool and refreshing, thanks to the terminal units working diligently behind the scenes. Terminal units, while often unseen and unheard, play a crucial role in maintaining a comfortable environment in various buildings, from offices to schools and hospitals. Let’s explore these components, the challenges they address, and how Dwyer’s range of products helps conquer these challenges.
Picture this: you walk into an office building on a hot summer day. As the door closes behind you, you immediately feel the temperature change. It’s cool and refreshing, thanks to the terminal units working diligently behind the scenes. Terminal units, while often unseen and unheard, play a crucial role in maintaining a comfortable environment in various buildings, from offices to schools and hospitals. Let’s explore these components, the challenges they address, and how Dwyer’s range of products helps conquer these challenges.
Continue reading “Exploring Terminal Units: Dwyer’s Solutions for Better Indoor Environments and Optimal Performance”
Reflections: AHR Expo, World of Concrete, and What’s Next!
The curtain has fallen on two fantastic tradeshows, AHR Expo and World of Concrete, and we’re still buzzing with excitement! We want to extend a heartfelt thank you to all the amazing customers, partners, and teammates who made these events unforgettable. Continue reading “Reflections: AHR Expo, World of Concrete, and What’s Next!”
Upcoming Tradeshows: Innovation at AHR Expo and World of Concrete

As the new year unfolds, Dwyer Instruments is gearing up to showcase its innovative solutions at two prominent tradeshows in January: AHR Expo and World of Concrete. These events provide a unique platform for professionals and enthusiasts to explore Dwyer’s latest offerings and engage in discussions about industry advancements.
Continue reading “Upcoming Tradeshows: Innovation at AHR Expo and World of Concrete”
On the Job with Dwyer: Series 490W Wireless Hydronic Differential Pressure Manometer

Dwyer’s Series 490W Wireless Hydronic Differential Pressure Manometer is the most accurate and easy-to-operate manometer on the market. Along with wireless transducers and a mobile device, a single operator using a 490W can monitor and balance a hydronic system in less time and with less manpower than would be possible using a traditional hydronic balancer. In addition, being wireless, the 490W has zero hoses or manifolds – meaning there are no annoying attachments that need to be carried, that can snag onto equipment, or that need to be drained between readings. Continue reading “On the Job with Dwyer: Series 490W Wireless Hydronic Differential Pressure Manometer”
Optimizing Grain Bin Systems: A Comprehensive Guide to Conveyor Systems and Dwyer Instruments
 Grain bin systems are the cornerstone of modern agricultural storage, offering a secure and efficient solution for managing harvested grain. Among the critical components of these systems, conveyor systems play a pivotal role in ensuring the smooth movement of grain within the bins. In this article, we will delve into the workings of various conveyor systems used with grain bins, providing insights into their functionality. Additionally, we will explore how Dwyer Instruments’ innovative products can enhance the monitoring and control of these conveyor systems. Continue reading “Optimizing Grain Bin Systems: A Comprehensive Guide to Conveyor Systems and Dwyer Instruments”
Grain bin systems are the cornerstone of modern agricultural storage, offering a secure and efficient solution for managing harvested grain. Among the critical components of these systems, conveyor systems play a pivotal role in ensuring the smooth movement of grain within the bins. In this article, we will delve into the workings of various conveyor systems used with grain bins, providing insights into their functionality. Additionally, we will explore how Dwyer Instruments’ innovative products can enhance the monitoring and control of these conveyor systems. Continue reading “Optimizing Grain Bin Systems: A Comprehensive Guide to Conveyor Systems and Dwyer Instruments”